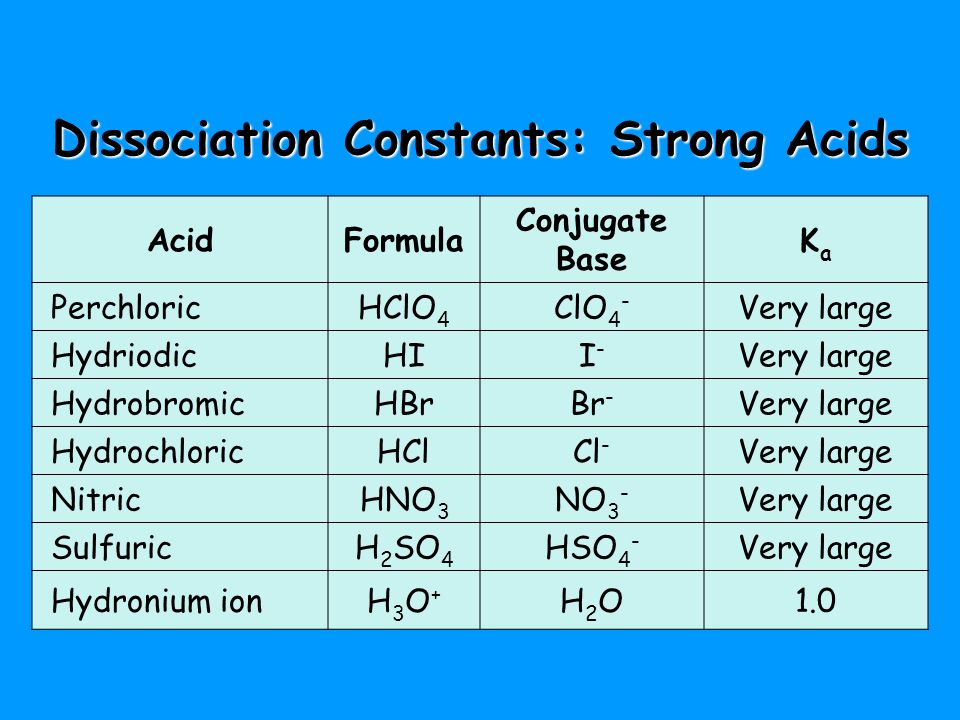
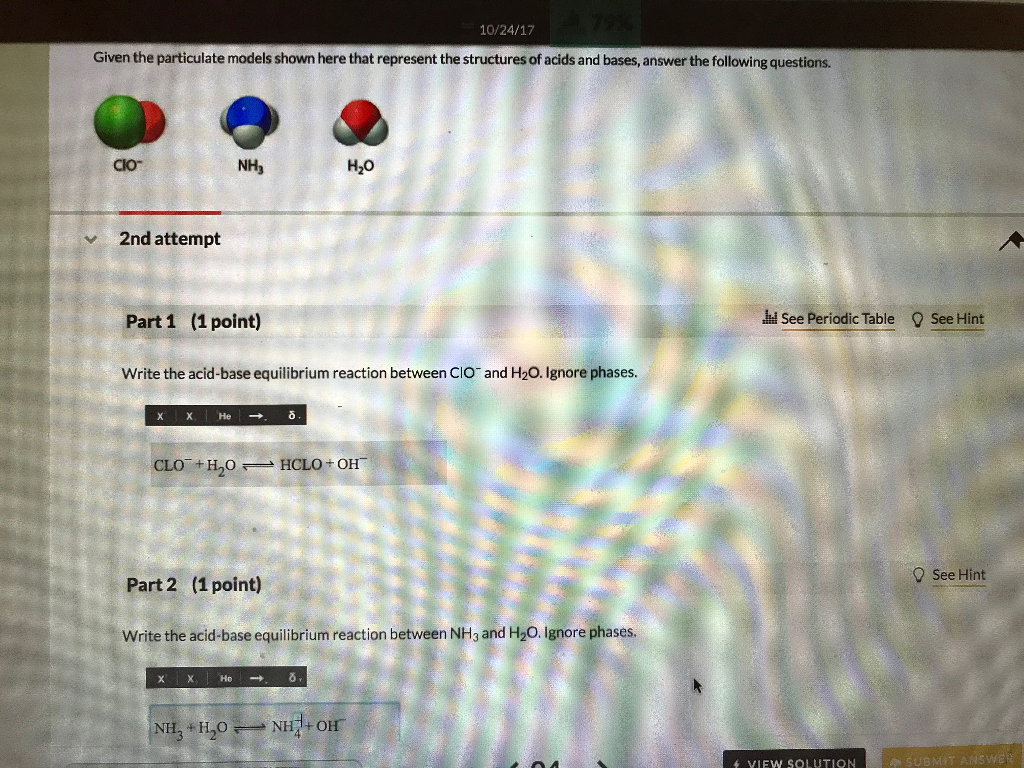
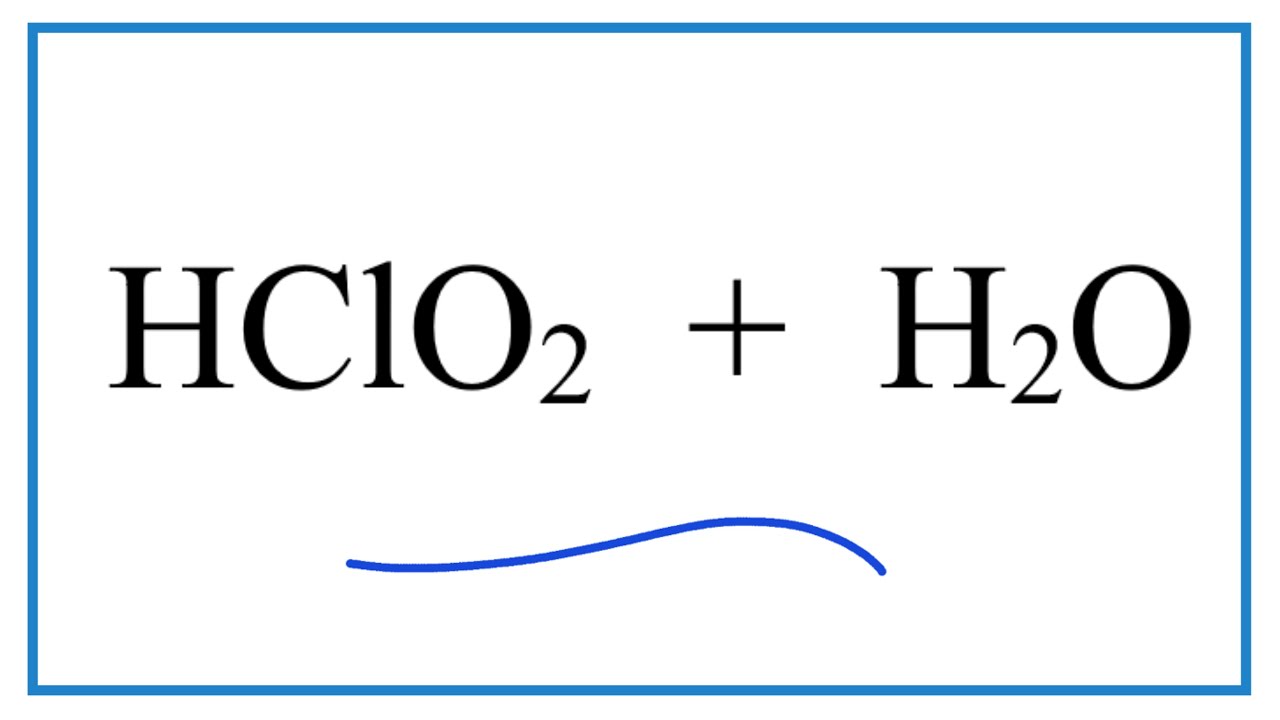OneClass: The chemical formula for the conjugate acid of ClO- is a. HClO- b. HClO c. Cl- d. no correc...

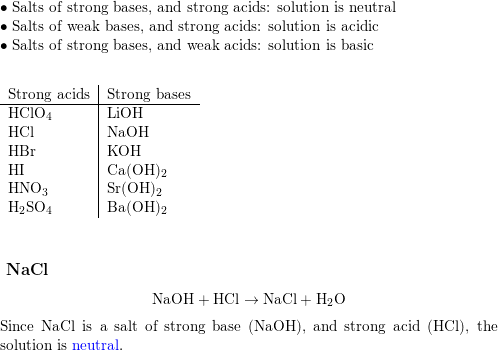
Reactions in Solution The most important substance on earth is water. In chemistry, water is necessary for many reactions to take place. Table salt (NaCl) - ppt download

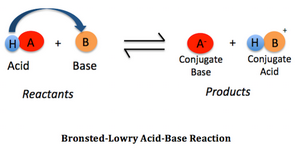
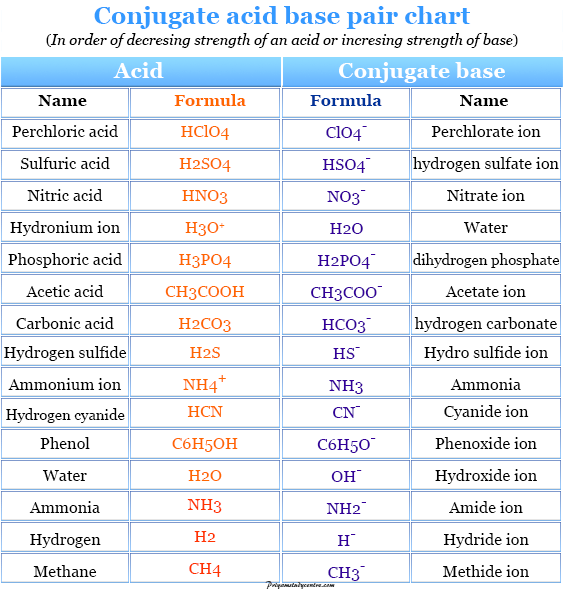
SOLVED: Identify the conjugate acid or base of each of the following: You have 3 attempts Conjugate base of HNOz HNO+ NO: HNO: NOz Conjugate base of HCLO HzClO HCl HClO2 ClO
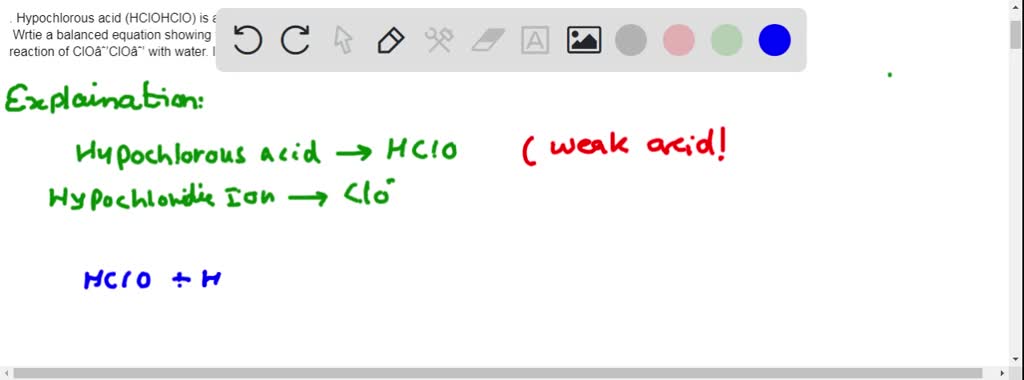
SOLVED: 1. Hypochlorous acid (HClOHClO) is a weak acid. The conjugate base of this acid is the hypochlorite ion (ClO−ClO−). Wrtie a balanced equation showing the reaction of HClOHClO with water. Include